Electrolysis. Electrolysis of melts and electrolyte solutions Obtaining hydrochloric acid by electrolysis
ELECTROLYSIS
melts and solutions of electrolytes
by electrolysis called the set of chemical reactions that occur during the passage of direct current through an electrochemical system consisting of two electrodes and an electrolyte melt or solution.
The chemical essence of electrolysis lies in the fact that it is a redox reaction occurring under the influence of a direct electric current, and the processes of oxidation and reduction are spatially separated.
Cathode - an electrode on which cations or water are reduced. It is negatively charged.
Anode - an electrode on which anions or water are oxidized. It is positively charged.
1. Electrolysis of melts of salts, bases.
During the electrolysis of melts, metal cations are always reduced at the cathode.
K(-): Men+ + nē → Me0
The anodic process is determined by the composition of the anion:
a) If the anion of an oxygen-free acid (Cl-, Br-, I-, S2-), then this anion undergoes anodic oxidation and a simple substance is formed:
A(+): 2Cl - - 2ē → Cl2 or A(+): S2- - 2ē → S0
b) If an oxygen-containing anion (SO42-, SiO32-, HO-, etc.) is subjected to anodic oxidation, then the non-metal forms an oxide (without changing its oxidation state) and oxygen is released.
A(+): 2SiO32-- 4ē → 2SiO2 + О2
A(+): 2SO32-- 4ē → 2SO2 + O2
A(+): 4РO43-- 12ē → 2Р2O5 + 3О2
A(+): 4NO3-- 4ē → 2N2O5 + О2
A(+): 4HO-- 4ē → 2H2O + O2
Example 1.1. Molten Salt ZnCl2
ZnCl2 Û Zn2+ + 2Cl-
S: ZnCl2 electrolysis of Zn + Cl2
Example 1.2. Alkali melt NaOH
NaOH Û Na+ + OH-
The total equation of electrolysis is obtained by adding the right and left parts of the equations, provided that the electrons involved in the cathode and anode processes are equal.
https://pandia.ru/text/80/299/images/image006_58.gif" width="70" height="12">4 Na+ + 4 ē + 4 OH - - 4 ē electrolysis 4 Na0 + O2 + 2H2O
4 Na+ + 4 OH - electrolysis 4 Na0 + O2 + 2H2O - ionic equation
4NaOH electrolysis 4Na + 2H2O + O2 - molecular equation
Example 1.3. Molten salt Na2SO4
Na2SO4 Û 2Na+ + SO42-
K(-): Na+ + 1 ē Þ Nao *4
A(+): 2SO42- - 4 ē Þ O2 + 2SO3
4Na+ +2SO42- Þ 2Nao + O2 + 2SO3 – ionic electrolysis equation
2Na2SO4 electrolysis 4Nao + O2 + 2SO3 - molecular equation
K A
Example 1.4. Molten salt AgNO3
AgNO3 Û Ag+ + NO3-
K(-): Ag+ + 1 ē Þ Ago *4
A(+): 4NO3- - 4 ē Þ 2N2O5 + 2O2 *1
4Ag+ + 4NO3- electrolysis 4Ag + 2N2O5 + 2O2
4AgNO3 electrolysis 4Ag + 2N2O5 +2O2
K A
Tasks for independent work . Compose equations for the electrolysis of melts of the following salts: AlCl3, Cr2(SO4)3, Na2SiO3, K2CO3.
2. Electrolysis of solutions of salts, hydroxides and acids.
The electrolysis of aqueous solutions is complicated by the fact that water can take part in the processes of oxidation and reduction.
cathodic processes determined by the electrochemical activity of the salt cation. The more to the left the metal is in the voltage series, the more difficult it is for its cations to be reduced at the cathode:
![]() Li
K
Ca
Na
mg
Al
Mn
Zn
Cr
Te
Ni
sn
Pb
H2
Cu
hg
Ag
Pt
Au
Li
K
Ca
Na
mg
Al
Mn
Zn
Cr
Te
Ni
sn
Pb
H2
Cu
hg
Ag
Pt
Au
I GroupIIGroupIIIGroup
For metal cations up to Al inclusive (group I), the cathodic process is the reduction of hydrogen from water:
(-)K: 2H2O + 2ē → H2 + 2HO-
For metal cations after hydrogen (group III), the cathodic process is their reduction to metal:
(-) K: Men+ + nē → Me0
For metal cations in the voltage series from Mn to H2 (group II), there are parallel competitive processes for the reduction of metal cations and hydrogen from water:
(-) K: Men+ + nē → Me0
2Н2О + 2ē → Н2+ 2НО-
Which of these processes will prevail depends on a number of factors: Me activity, solution pH, salt concentration, applied voltage, and electrolysis conditions.
Anode processes are determined by the composition of salt anions:
a) If the anion is an oxygen-free acid (Cl-, Br-, I-, S2-, etc.), then it is oxidized to simple substances (with the exception of F-):
A(+): S2- - 2ē → S0
b) In the presence of an oxygen-containing anion (SO42-, CO32-, etc. or OH-), only water undergoes anodic oxidation:
A(+): 2H2O - 4ē → O2 + 4H+
Consider examples illustrating all possible options:
Example 2.1 . KCl salt solution
K(-): 2H2O + 2e - Þ H2 + 2OH-
A(+): 2Cl - - 2e - Þ Cl2
å: 2H2O + 2Cl - electrolysis H2 + 2OH - + Cl2 - ionic electrolysis equation
2KCl + 2H2O electrolysis H2 + 2KOH + Cl2 - electrolysis molecular equation
K A
Example 2.2 . CuCl2 salt solution
CuCl2 Û Cu2+ + 2Cl-
K(-): Cu2+ + 2e - Þ Cuo
A(+): 2Cl- -2e - Þ Cl2
å: CuCl2 electrolysis of Cu + Cl2
Example 2.3. FeCl2 salt solution
FeCl2 Û Fe2+ + 2Cl-
Iron belongs to group II metals, so two parallel processes will take place on the cathode:
1st process:
(-) K: Fe2+ + 2ē → Fe0
(+)A: 2Cl - - 2ē → Cl2
Fe2+ + 2Cl - el-z Fe0 + Cl2 - ionic equation of the process
FeCl2 el-z Fe0 + Cl2 - molecular equation of the process
2nd process:
(-)K: 2H2O + 2ē → H2+ 2OH-
(+)A: 2Cl - - 2ē → Cl2
2H2O + 2Cl - → H2+ 2OH - + Cl2 - ionic equation of the process
2H2O + FeCl2 electrolysis of H2 + Fe(OH)2 + Cl2 - molecular equation.
In this way, in the cathode space Fe, H2 and Fe(OH)2 will be formed in different ratios depending on the electrolysis conditions.
Example 2.4 . Salt solution Na2SO4.
Na2SO4 Û 2Na+ + SO42-
K(-) 2H2O + 2e - Þ H2 + 2OH - *2
A(+) 2H2O – 4e - Þ O2 + 4H+
å: 6H2O electrolysis 2H2 + 4OH - + O2 + 4H+
å: 6H2O + 2Na2SO4 electrolysis 2H2 + 4 NaOH + O2 + 2H2SO4
in the cathode space in the anode space
When the electric current is turned off and the contents of the cathode and anode space are mixed, the final result of electrolysis can be represented by the scheme:
2H2O el-z 2H2 + O2,
since the alkali will react with the acid to form 2 mol of salt and 4 mol of water.
Example 2.5 . Electrolysis of CuSO4 solution.
CuSO4 Û Cu2+ + SO42-
K(-): Cu2+ + 2e - Þ Cuo
A(+): 2H2O – 4e - Þ O2 + 4H+
å: 2Cu2+ + 2H2O electrolysis 2Cuo + O2 + 4H+
å: CuSO4 + 2H2O electrolysis 2Cuo + O2 + 2H2SO4
Example 2.6. Electrolysis of FeSO4 solution
Since iron belongs to group II metals, two competitive processes will go on in parallel at the cathode (see example 2.3), and water will be oxidized at the anode (see example 2.4):
1st process:
https://pandia.ru/text/80/299/images/image043_10.gif" width="41" height="12">2Fe2+ + 2Н2О el-z 2 Fe + O2 + 4H+ - ionic equation of the process
2FeSO4 + 2Н2О el-z 2 Fe + O2 + 2Н2SO4– molecular equation
2nd process:
K(+): 2H2O + 2ē → H2+ 2OH - *2
A(-): 2H2O - 4ē → O2+ 4H+
6H2O electrolysis 2H2+ 4OH - + O2 + 4H+
6Н2О + 2FeSO4 electrolysis 2Н2+ 2Fe(OH)2 + O2 + 2Н2SO4 - molecular
https://pandia.ru/text/80/299/images/image051_9.gif" width="21" height="50">And only if the processes of cathodic reduction of metal and hydrogen cations from water are in equal proportions , we can write the total final equation of the reaction:
(-) K: Fe2+ + 2ē → Fe0
2H2O + 2ē → H2+ 2HO - only 4 electrons
(+)A: 2H2O - 4ē → O2 + 4H+
Fe2+ + 2H2O + 2H2O → Fe + H2+ 2HO - + O2 + 4H+
2FeSO4 + 4Н2О el-z Fe + Н2+ Fe(OH)2 + O2 + 2Н2SO4
cathode anode
After turning off the current and mixing the solutions, the final equation will be as follows:
· Write electrolysis equations for K2CO3, ZnSO4, AgNO3, NiI2, CoCl2 solutions.
· To solve the task. To analyze the content of NaCl impurity in technical NaOH, 40 g of the drug was dissolved in water and subjected to electrolysis until complete oxidation of chlorine ions. In this case, 601 ml of Cl2 were released at the anode at a temperature of 200C and normal pressure. Calculate mass fraction NaCl impurities in NaOH.
3. Electrolysis c soluble s m anode
Above were considered examples of the electrolysis of aqueous solutions of salts with an inert anode, i.e. one that does not take a chemical part in the anode process. Such electrodes are made of inactive noble metals, such as Pt, Ir, or carbon electrodes are used. If soluble anodes are used, for example, Cu-anode, Zn-anode, then the anode process changes significantly, since the anode itself is oxidized. At the anode of the 2 competitive ones, a process with a lower potential is underway: for the oxidation of copper, E0 = - 0.34 V, for the oxidation of zinc, E0 = - 0.76 V, and for the oxidation of the Cl anion, E0 = + 1.36 V.
Example 3.1. Electrolysis of an aqueous solution of CuCl2 salt with a soluble anode:
Cathode (-): Cu-anode (+):
Сu2+ + 2ē → Cu0 Сu0 - 2ē → Cu2+
Thus, a kind of refining of the copper anode occurs: it dissolves, impurities remain in the anode space, and pure copper is deposited on the cathode. Chlorine anion is not oxidized, but accumulates in the anode space.
Example 3.2. Electrolysis of an aqueous solution of KCl salt with a Cu anode:
Cu-anode (+): Сu0 - 2ē → Cu2+
At the cathode, at the initial moment, hydrogen begins to be reduced from water, but the appearance of Cu2+ in solution makes two cathodic reduction reactions competitive:
K (-): 2H2O + 2ē → H2 + 2HO - E0 \u003d - 0.828 V
Cu2+ + 2ē → Cu0 E0 = + 0.34 V
As a result, the one that is characterized by a higher potential proceeds predominantly, i.e., the reduction of Сu2+ to Cu0.
Thus, in this case, the dissolution of the Cu-anode will occur: Сu0 - 2ē → Cu2+, and the copper cations formed on the cathode will be reduced: Сu2+ + 2ē → Cu0. Salt KCl is needed only to increase the electrical conductivity of the solution, and it does not take a direct part in redox processes.
Assignment for independent work. Consider the electrolysis of CuSO4 with a Cu anode, Na2SO4 with a Cu anode.
Solution electrolysis
and molten salts (2 hours)
Classes of the elective course "Electrochemistry"
Goals of the first lesson:
First lesson plan
1. Repetition of the studied methods for obtaining metals.
2. Explanation of new material.
3. Solving problems from the textbook by G.E. Rudzitis, F.G. Feldman "Chemistry-9" (M .: Education, 2002), p. 120, no. 1, 2.
4. Checking the assimilation of knowledge on test tasks.
5. Report on the application of electrolysis.
Goals of the first lesson: to teach how to write schemes for the electrolysis of solutions and molten salts and apply the knowledge gained to solve calculation problems; continue the formation of skills in working with a textbook, test materials; discuss the application of electrolysis in national economy.
PROGRESS OF THE FIRST LESSON
Repetition of learned methods obtaining metals on the example of obtaining copper from copper(II) oxide.
Recording the equations of the corresponding reactions:
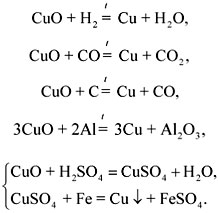
Another way to obtain metals from solutions and melts of their salts is electrochemical, or electrolysis.
Electrolysis is a redox process that occurs on electrodes when an electric current is passed through a melt or electrolyte solution..
Electrolysis of sodium chloride melt:
NaCl Na + + Cl – ;
cathode (–) (Na +): Na + + e= Na 0 ,
anode (–) (Cl –): Cl – – e\u003d Cl 0, 2Cl 0 \u003d Cl 2;
2NaCl \u003d 2Na + Cl 2.
Electrolysis of sodium chloride solution:
NaCl Na + + Cl – ,
H 2 O H + + OH -;
cathode (–) (Na +; H +): H + + e= H 0 , 2H 0 = H 2
(2H 2 O + 2 e\u003d H 2 + 2OH -),
anode (+) (Cl - ; OH -): Cl - - e\u003d Cl 0, 2Cl 0 \u003d Cl 2;
2NaCl + 2H 2 O \u003d 2NaOH + Cl 2 + H 2.
Electrolysis of copper(II) nitrate solution:
Cu(NO 3) 2 Cu 2+ +
H 2 O H + + OH -;
cathode (–) (Cu 2+; H +): Cu 2+ + 2 e= Cu 0 ,
anode (+) (OH -): OH - - e=OH0,
4H 0 \u003d O 2 + 2H 2 O;
2Cu(NO 3) 2 + 2H 2 O \u003d 2Cu + O 2 + 4HNO 3.
These three examples show why it is more profitable to carry out electrolysis than to carry out other methods of obtaining metals: metals, hydroxides, acids, gases are obtained.
We wrote the electrolysis schemes, and now we will try to write the electrolysis equations right away, without referring to the schemes, but only using the ion activity scale:
Examples of electrolysis equations:
2HgSO 4 + 2H 2 O \u003d 2Hg + O 2 + 2H 2 SO 4;
Na 2 SO 4 + 2H 2 O \u003d Na 2 SO 4 + 2H 2 + O 2;
2LiCl + 2H 2 O \u003d 2LiOH + H 2 + Cl 2.
Problem solving from the textbook by G.E. Rudzitis and F.G. Feldman (9th grade, p. 120, No. 1, 2).
Task 1. During the electrolysis of a solution of copper (II) chloride, the mass of the cathode increased by 8 g. What gas was released, what is its mass?
Solution
CuCl 2 + H 2 O \u003d Cu + Cl 2 + H 2 O,
(Cu) \u003d 8/64 \u003d 0.125 mol,
(Cu) \u003d (Сl 2) \u003d 0.125 mol,
m(Cl 2) \u003d 0.125 71 \u003d 8.875 g.
Answer. The gas is chlorine with a mass of 8.875 g.
Task 2. During the electrolysis of an aqueous solution of silver nitrate, 5.6 liters of gas were released. How many grams of metal deposited on the cathode?
Solution
4AgNO 3 + 2H 2 O \u003d 4Ag + O 2 + 4HNO 3,
(O 2) \u003d 5.6 / 22.4 \u003d 0.25 mol,
(Ag) \u003d 4 (O 2) \u003d 4 25 \u003d 1 mol,
m(Ag) \u003d 1 107 \u003d 107 g.
Answer. 107 g of silver.
Testing
Option 1
1. During the electrolysis of a potassium hydroxide solution at the cathode, the following is released:
a) hydrogen; b) oxygen; c) potassium.
2. During the electrolysis of a solution of copper(II) sulfate in solution, the following is formed:
a) copper(II) hydroxide;
b) sulfuric acid;
3. During the electrolysis of a solution of barium chloride at the anode, the following is released:
a) hydrogen; b) chlorine; c) oxygen.
4. During the electrolysis of an aluminum chloride melt, the following is released at the cathode:
a) aluminum; b) chlorine;
c) electrolysis is impossible.
5. The electrolysis of a solution of silver nitrate proceeds according to the following scheme:
a) AgNO 3 + H 2 O Ag + H 2 + HNO 3;
b) AgNO 3 + H 2 O Ag + O 2 + HNO 3;
c) AgNO 3 + H 2 O AgNO 3 + H 2 + O 2.
Option 2
1. During the electrolysis of a sodium hydroxide solution at the anode, the following is released:
a) sodium; b) oxygen; c) hydrogen.
2. During the electrolysis of a solution of sodium sulfide in solution, the following is formed:
a) hydrosulphuric acid;
b) sodium hydroxide;
3. During the electrolysis of a mercury(II) chloride melt, the following is released at the cathode:
a) mercury; b) chlorine; c) electrolysis is impossible.
4.
5. The electrolysis of a solution of mercury(II) nitrate proceeds according to the following scheme:
a) Hg (NO 3) 2 + H 2 O Hg + H 2 + HNO 3;
b) Hg (NO 3) 2 + H 2 O Hg + O 2 + HNO 3;
c) Hg (NO 3) 2 + H 2 O Hg (NO 3) 2 + H 2 + O 2.
Option 3
1. During the electrolysis of a solution of copper (II) nitrate, the following is released at the cathode:
a) copper; b) oxygen; c) hydrogen.
2. During the electrolysis of a solution of lithium bromide in solution, the following is formed:
b) hydrobromic acid;
c) lithium hydroxide.
3. During the electrolysis of a silver chloride melt, the following is released at the cathode:
a) silver; b) chlorine; c) electrolysis is impossible.
4. During the electrolysis of an aluminum chloride solution, aluminum is released into:
a) cathode; b) anode; c) remains in solution.
5. The electrolysis of a solution of barium bromide proceeds according to the following scheme:
a) BaBr 2 + H 2 O Br 2 + H 2 + Ba (OH) 2;
b) BaBr 2 + H 2 O Br 2 + Ba + H 2 O;
c) BaBr 2 + H 2 O Br 2 + O 2 + Ba (OH) 2.
Option 4
1. During the electrolysis of a barium hydroxide solution at the anode, the following is released:
a) hydrogen; b) oxygen; c) barium.
2. During the electrolysis of a solution of potassium iodide in solution, the following is formed:
a) hydroiodic acid;
b) water; c) potassium hydroxide.
3. During the electrolysis of a melt of lead (II) chloride, the following is released at the cathode:
a) lead; b) chlorine; c) electrolysis is impossible.
4. During the electrolysis of a silver nitrate solution at the cathode, the following is released:
a) silver; b) hydrogen; c) oxygen.
5. The electrolysis of sodium sulfide solution proceeds according to the following scheme:
a) Na 2 S + H 2 O S + H 2 + NaOH;
b) Na 2 S + H 2 O H 2 + O 2 + Na 2 S;
c) Na 2 S + H 2 O H 2 + Na 2 S + NaOH.
Answers
| Option | Question 1 | Question 2 | Question 3 | Question 4 | Question 5 |
| 1 | a | b | b | a | b |
| 2 | b | b | a | a | b |
| 3 | a | in | a | in | a |
| 4 | b | in | a | a | a |
The use of electrolysis in the national economy
1. To protect metal products from corrosion, a thin layer of another metal is applied to their surface: chromium, silver, gold, nickel, etc. Sometimes, in order not to waste expensive metals, a multi-layer coating is produced. For example, the exterior parts of a car are first covered with a thin layer of copper, a thin layer of nickel is applied to the copper, and a layer of chromium is applied to it.
When applying coatings to metal by electrolysis, they are obtained even in thickness and durable. In this way, you can cover products of any shape. This branch of applied electrochemistry is called electroplating.
2. In addition to corrosion protection, galvanic coatings give a beautiful decorative look to products.
3. Another branch of electrochemistry, close in principle to electroplating, is called electroplating. This is the process of obtaining exact copies of various items. To do this, the object is covered with wax and a matrix is obtained. All recesses of the copied object on the matrix will be bulges. The surface of the wax matrix is coated with a thin layer of graphite, making it electrically conductive.
The resulting graphite electrode is immersed in a bath of copper sulfate solution. The anode is copper. During electrolysis, the copper anode dissolves, and copper is deposited on the graphite cathode. Thus, an exact copper copy is obtained.
With the help of electroforming, clichés for printing, gramophone records are made, various objects are metallized. Galvanoplasty was discovered by the Russian scientist B.S. Jacobi (1838).
Making record dies involves applying a thin layer of silver to a plastic record to make it electrically conductive. Then an electrolytic nickel coating is applied to the plate.
What should be done to make a plate in an electrolytic bath - anode or cathode?
(About the e t. Cathode.)
4. Electrolysis is used to obtain many metals: alkali, alkaline earth, aluminum, lanthanides, etc.
5. To clean some metals from impurities, the metal with impurities is connected to the anode. The metal is dissolved during the electrolysis process and precipitated on the metal cathode, while the impurity remains in solution.
6. Electrolysis is widely used to obtain complex substances (alkalis, oxygen-containing acids), halogens.
Practical work(second lesson)
Lesson goals. Conduct water electrolysis, show electroplating in practice, consolidate the knowledge gained in the first lesson.
Equipment.On student tables: a flat battery, two wires with terminals, two graphite electrodes, a beaker, test tubes, a tripod with two legs, 3% sodium sulfate solution, a spirit lamp, matches, a torch.
On the teacher's desk: the same + a solution of copper sulfate, a brass key, a copper tube (a piece of copper).
Student briefing
1. Attach the wires with terminals to the electrodes.
2. Place the electrodes in a glass so that they do not touch.
3. Pour the electrolyte solution (sodium sulfate) into the beaker.
4. Pour water into the test tubes and, putting them upside down in a glass with electrolyte, put them on the graphite electrodes one by one, fixing the upper edge of the test tube in the foot of the tripod.
5. After the device is mounted, attach the ends of the wires to the battery.
6. Observe the evolution of gas bubbles: less of them are released at the anode than at the cathode. After almost all the water in one test tube is displaced by the released gas, and in the other - by half, disconnect the wires from the battery.
7. Light the spirit lamp, carefully remove the test tube, where the water is almost completely displaced, and bring it to the spirit lamp - a characteristic pop of gas will be heard.
8. Light a torch. Remove the second test tube, check with a smoldering splint of gas.
Assignments for students
1. Sketch the device.
2. Write an equation for the electrolysis of water and explain why it was necessary to carry out electrolysis in a solution of sodium sulfate.
3. Write reaction equations that reflect the release of gases on the electrodes.
Teacher demonstration experiment
(can be performed by the best students in the class
with appropriate equipment)
1. Connect the wire terminals to the copper tube and brass key.
2. Lower the tube and key into a beaker with copper(II) sulfate solution.
3. Connect the second ends of the wires to the battery: "minus" of the battery to the copper tube, "plus" to the key!
4. Observe the release of copper on the surface of the key.
5. After performing the experiment, first disconnect the terminals from the battery, then remove the key from the solution.
6. Disassemble the electrolysis circuit with a soluble electrode:
CuSO 4 \u003d Cu 2+ +
anode (+): Сu 0 - 2 e\u003d Cu 2+,
cathode (–): Cu 2+ + 2 e= Сu 0 .
The overall equation for electrolysis with a soluble anode cannot be written.
The electrolysis was carried out in a solution of copper(II) sulfate, because:
a) an electrolyte solution is needed in order for an electric current to flow, tk. water is a weak electrolyte;
b) no by-products of the reactions will be released, but only copper at the cathode.
7. To consolidate the past, write a scheme for the electrolysis of zinc chloride with carbon electrodes:
ZnCl 2 \u003d Zn 2+ + 2Cl -,
cathode (–): Zn 2+ + 2 e= Zn 0 ,
2H2O+2 e\u003d H 2 + 2OH -,
anode (+): 2Cl – – 2 e=Cl2.
The overall reaction equation in this case cannot be written, because it is not known what part of the total amount of electricity goes to the reduction of water, and what part - to the reduction of zinc ions.
 |
Scheme of the demonstration experiment |
Homework
1. Write an equation for the electrolysis of a solution containing a mixture of copper(II) nitrate and silver nitrate with inert electrodes.
2. Write the equation for the electrolysis of sodium hydroxide solution.
3. To clean a copper coin, it must be hung on a copper wire connected to the negative pole of the battery, and lowered into a 2.5% NaOH solution, where the graphite electrode connected to the positive pole of the battery should also be immersed. Explain how a coin becomes clean. ( Answer. Hydrogen ions are being reduced at the cathode:
2H + + 2 e\u003d H 2.
Hydrogen reacts with copper oxide on the surface of the coin:
CuO + H 2 \u003d Cu + H 2 O.
This method is better than powder cleaning, because. the coin is not erased.)
ELECTROLYSIS
One of the ways to obtain metals is electrolysis. Active metals occur in nature only in the form of chemical compounds. How to isolate from these compounds in the free state?
Solutions and melts of electrolytes conduct electric current. However, when current is passed through an electrolyte solution, chemical reactions can occur. Consider what will happen if two metal plates are placed in an electrolyte solution or melt, each of which is connected to one of the poles of the current source. These plates are called electrodes. Electric current is a moving stream of electrons. As a result of the fact that the electrons in the circuit move from one electrode to another, an excess of electrons appears on one of the electrodes. The electrons have a negative charge, so this electrode becomes negatively charged. It is called the cathode. On the other electrode, a lack of electrons is created, and it is positively charged. This electrode is called the anode. An electrolyte in a solution or melt dissociates into positively charged ions - cations and negatively charged ions - anions. Cations are attracted to a negatively charged electrode - the cathode. Anions are attracted to a positively charged electrode - the anode. On the surface of the electrodes, interaction between ions and electrons can occur.
Electrolysis refers to the processes that occur when an electric current is passed through solutions or melts of electrolytes.
The processes occurring during the electrolysis of solutions and melts of electrolytes are quite different. Let's consider both of these cases in detail.
Melt electrolysis
As an example, consider the electrolysis of a sodium chloride melt. In the melt, sodium chloride dissociates into ions Na+
and Cl - : NaCl = Na + + Cl -
Sodium cations move to the surface of a negatively charged electrode - the cathode. There is an excess of electrons on the cathode surface. Therefore, there is a transfer of electrons from the electrode surface to sodium ions. At the same time, ions Na+ are converted into sodium atoms, that is, cations are reduced Na+ . Process equation:
Na + + e - = Na
Chloride ions Cl - move to the surface of a positively charged electrode - the anode. A lack of electrons is created on the anode surface and electrons are transferred from anions Cl- to the surface of the electrode. At the same time, negatively charged ions Cl- are converted into chlorine atoms, which immediately combine to form chlorine molecules C l2 :
2C l - -2e - \u003d Cl 2
Chloride ions lose electrons, that is, they are oxidized.
Let us write together the equations of the processes occurring at the cathode and anode
Na + + e - = Na
2 C l - -2 e - \u003d Cl 2
One electron is involved in the process of reduction of sodium cations, and 2 electrons are involved in the process of oxidation of chlorine ions. However, the law of conservation of electric charge must be observed, that is, the total charge of all particles in the solution must be constant. Therefore, the number of electrons involved in the reduction of sodium cations must be equal to the number of electrons involved in the oxidation of chloride ions. Therefore, we multiply the first equation by 2:
Na + + e - \u003d Na 2
2C l - -2e - \u003d Cl 2 1
We add both equations together and get the general equation for the reaction.
2 Na + + 2C l - \u003d 2 Na + Cl 2 (ionic reaction equation), or
2 NaCl \u003d 2 Na + Cl 2 (molecular reaction equation)
So, in the considered example, we see that electrolysis is a redox reaction. At the cathode, the reduction of positively charged ions - cations, at the anode - the oxidation of negatively charged ions - anions. To remember which process happens where, you can use the "T rule":
cathode - cation - reduction.
Example 2Electrolysis of sodium hydroxide melt.
Sodium hydroxide in solution dissociates into cations and hydroxide ions.
Cathode (-)<-- Na + + OH - à Анод (+)
On the cathode surface, sodium cations are reduced, and sodium atoms are formed:
cathode (-) Na + +e à Na
Hydroxide ions are oxidized on the anode surface, while oxygen is released and water molecules are formed:
cathode (-) Na + + e à Na
anode (+)4 OH - - 4 e à 2 H 2 O + O 2
The number of electrons involved in the reduction reaction of sodium cations and in the oxidation reaction of hydroxide ions should be the same. So let's multiply the first equation by 4:
cathode (-) Na + + e à Na 4
anode (+)4 OH - – 4 e à 2 H 2 O + O 2 1
Putting both equations together, we get the equation for the electrolysis reaction:
4 NaOH à 4 Na + 2 H 2 O + O 2
Example 3Consider the electrolysis of the melt Al2O3
Using this reaction, aluminum is obtained from bauxite, a natural compound that contains a lot of aluminum oxide. The melting point of aluminum oxide is very high (more than 2000º C), so special additives are added to it, lowering the melting point to 800-900º C. In the melt, aluminum oxide dissociates into ions Al 3+ and O 2-. H cations are reduced at the cathode Al 3+ , turning into aluminum atoms:
Al +3 e a Al
Anions are oxidized at the anode O 2- turning into oxygen atoms. Oxygen atoms immediately combine into O 2 molecules:
2 O 2- – 4 e à O 2
The number of electrons involved in the reduction of aluminum cations and the oxidation of oxygen ions must be equal, so we multiply the first equation by 4, and the second by 3:
Al 3+ +3 e à Al 0 4
2 O 2- – 4 e à O 2 3
Let's add both equations and get
4 Al 3+ + 6 O 2- a 4 Al 0 +3 O 2 0 (ionic reaction equation)
2 Al 2 O 3 à 4 Al + 3 O 2
Solution electrolysis
In the case of passing an electric current through an aqueous electrolyte solution, the matter is complicated by the presence of water molecules in the solution, which can also interact with electrons. Recall that in a water molecule, hydrogen and oxygen atoms are connected by a polar covalent bond. The electronegativity of oxygen is greater than the electronegativity of hydrogen, so the shared electron pairs are shifted towards the oxygen atom. A partial negative charge arises on the oxygen atom, it is denoted δ-, and on hydrogen atoms it has a partial positive charge, it is denoted δ+.
δ+
H-O δ-
│
H δ+
Due to this shift of charges, the water molecule has positive and negative "poles". Therefore, water molecules can be attracted by a positively charged pole to a negatively charged electrode - the cathode, and by a negative pole - to a positively charged electrode - anode. At the cathode, water molecules can be reduced, and hydrogen is released:
Oxidation of water molecules can occur at the anode with the release of oxygen:
2 H 2 O - 4e - \u003d 4H + + O 2
Therefore, either electrolyte cations or water molecules can be reduced at the cathode. These two processes seem to compete with each other. What process actually takes place at the cathode depends on the nature of the metal. Whether metal cations or water molecules will be reduced at the cathode depends on the position of the metal in series of metal stresses .
Li K Na Ca Mg Al ¦¦ Zn Fe Ni Sn Pb (H 2) ¦¦ Cu Hg Ag Au
If the metal is in the voltage series to the right of hydrogen, metal cations are reduced at the cathode and free metal is released. If the metal is in the voltage series to the left of aluminum, water molecules are reduced at the cathode and hydrogen is released. Finally, in the case of metal cations from zinc to lead, either metal evolution or hydrogen evolution can occur, and sometimes both hydrogen and metal are evolved simultaneously. In general, this is a rather complicated case, much depends on the reaction conditions: the concentration of the solution, the current strength, and others.
One of two processes can also occur at the anode - either the oxidation of electrolyte anions, or the oxidation of water molecules. Which process actually takes place depends on the nature of the anion. During the electrolysis of salts of anoxic acids or the acids themselves, anions are oxidized at the anode. The only exception is the fluoride ion F- . In the case of oxygen-containing acids, water molecules are oxidized at the anode and oxygen is released.
Example 1Let's look at the electrolysis of an aqueous solution of sodium chloride.
In an aqueous solution of sodium chloride there will be sodium cations Na + , chlorine anions Cl - and water molecules.
2 NaCl a 2 Na + + 2 Cl -
2Н 2 О а 2 H + + 2 OH -
cathode (-) 2 Na + ; 2 H + ; 2Н + + 2е а Н 0 2
anode (+) 2 Cl - ; 2OH-; 2 Cl - – 2e a 2 Cl 0
2NaCl + 2H 2 O à H 2 + Cl 2 + 2NaOH
Chemical activity anions hardly decreases.
Example 2What if the salt contains SO 4 2- ? Consider the electrolysis of a nickel sulfate solution ( II ). nickel sulfate ( II ) dissociates into ions Ni 2+ and SO 4 2-:
NiSO 4 à Ni 2+ + SO 4 2-
H 2 O à H + + OH -
Nickel cations are between metal ions Al 3+ and Pb 2+ , occupying a middle position in the voltage series, the recovery process at the cathode occurs according to both schemes:
2 H 2 O + 2e - \u003d H 2 + 2OH -
Anions of oxygen-containing acids are not oxidized at the anode ( anion activity series ), water molecules are oxidized:
anode e à O 2 + 4H +
Let us write together the equations of the processes occurring at the cathode and anode:
cathode (-) Ni 2+ ; H + ; Ni 2+ + 2е а Ni 0
2 H 2 O + 2e - \u003d H 2 + 2OH -
anode (+) SO 4 2- ; OH -; 2H 2 O - 4 e à O 2 + 4H +
4 electrons are involved in the reduction processes, and 4 electrons are also involved in the oxidation process. Putting these equations together, we get the general reaction equation:
Ni 2+ +2 H 2 O + 2 H 2 O à Ni 0 + H 2 + 2OH - + O 2 + 4 H +
On the right side of the equation, there are simultaneously H + ions and oh- , which combine to form water molecules:
H + + OH - à H 2 O
Therefore, on the right side of the equation, instead of 4 H + ions and 2 ions oh- we write 2 water molecules and 2 H + ions:
Ni 2+ +2 H 2 O + 2 H 2 O à Ni 0 + H 2 +2 H 2 O + O 2 + 2 H +
Let's reduce two water molecules on both sides of the equation:
Ni 2+ +2 H 2 O à Ni 0 + H 2 + O 2 + 2 H +
This is a short ionic equation. To get the full ionic equation, you need to add to both parts of the sulfate ion SO 4 2- , formed during the dissociation of nickel sulfate ( II ) and not participating in the reaction:
Ni 2+ + SO 4 2- + 2H 2 O à Ni 0 + H 2 + O 2 + 2H + + SO 4 2-
Thus, during the electrolysis of a solution of nickel sulfate ( II ) hydrogen and nickel are released at the cathode, and oxygen is released at the anode.
NiSO 4 + 2H 2 O à Ni + H 2 + H 2 SO 4 + O 2
Example 3 Write the equations of the processes occurring during the electrolysis of an aqueous solution of sodium sulfate with an inert anode.
Standard electrode potential of the system Na + + e = Na 0 is much more negative than the potential of the water electrode in a neutral aqueous medium (-0.41 V). Therefore, electrochemical reduction of water will occur on the cathode, accompanied by hydrogen evolution
2Н 2 О а 2 H + + 2 OH -
and Na ions + coming to the cathode will accumulate in the adjacent part of the solution (cathode space).
At the anode, electrochemical oxidation of water will occur, leading to the release of oxygen.
2 H 2 O - 4e à O 2 + 4 H +
because corresponding to this system standard electrode potential (1.23 V) is significantly lower than the standard electrode potential (2.01 V) that characterizes the system
2 SO 4 2- + 2 e \u003d S 2 O 8 2-.
Ions SO 4 2- moving towards the anode during electrolysis will accumulate in the anode space.
Multiplying the equation of the cathode process by two, and adding it with the equation of the anode process, we obtain the total equation of the electrolysis process:
6 H 2 O \u003d 2 H 2 + 4 OH - + O 2 + 4 H +
Taking into account that ions are simultaneously accumulated in the cathode space and ions in the anode space, the overall process equation can be written in the following form:
6H 2 O + 2Na 2 SO 4 \u003d 2H 2 + 4Na + + 4OH - + O 2 + 4H + + 2SO 4 2-
Thus, simultaneously with the release of hydrogen and oxygen, sodium hydroxide (in the cathode space) and sulfuric acid (in the anode space) are formed.
Example 4Electrolysis of copper sulfate solution ( II) CuSO4.
Cathode (-)<-- Cu 2+ + SO 4 2- à анод (+)
cathode (-) Cu 2+ + 2e à Cu 0 2
anode (+) 2H 2 O - 4 e à O 2 + 4H + 1
H + ions remain in the solution and SO 4 2- , since sulfuric acid accumulates.
2CuSO 4 + 2H 2 O à 2Cu + 2H 2 SO 4 + O 2
Example 5 Electrolysis of copper chloride solution ( II) CuCl 2 .
Cathode (-)<-- Cu 2+ + 2Cl - à анод (+)
cathode (-) Cu 2+ + 2e à Cu 0
anode (+) 2Cl - – 2e à Cl 0 2
Both equations involve two electrons.
Cu 2+ + 2e à Cu 0 1
2Cl - -– 2e à Cl 2 1
Cu 2+ + 2 Cl - à Cu 0 + Cl 2 (ionic equation)
CuCl 2 à Cu + Cl 2 (molecular equation)
Example 6 Electrolysis of silver nitrate solution AgNO3.
Cathode (-)<-- Ag + + NO 3 - à Анод (+)
cathode (-) Ag + + e à Ag 0
anode (+) 2H 2 O - 4 e à O 2 + 4H +
Ag + + e à Ag 0 4
2H 2 O - 4 e à O 2 + 4H + 1
4 Ag + + 2 H 2 O à 4 Ag 0 + 4 H + + O 2 (ionic equation)
4 Ag + + 2 H 2 Oà 4 Ag 0 + 4 H + + O 2 + 4 NO 3 - (full ionic equation)
4 AgNO 3 + 2 H 2 Oà 4 Ag 0 + 4 HNO 3 + O 2 (molecular equation)
Example 7 Electrolysis of hydrochloric acid solutionHCl.
Cathode (-)<-- H + + Cl - à anode (+)
cathode (-) 2H + + 2 eà H 2
anode (+) 2Cl - – 2 eà Cl 2
2 H + + 2 Cl - à H 2 + Cl 2 (ionic equation)
2 HClà H 2 + Cl 2 (molecular equation)
Example 8 Electrolysis of sulfuric acid solutionH 2 SO 4 .
Cathode (-) <-- 2H + + SO 4 2- à anode (+)
cathode (-)2H+ + 2eà H2
anode(+) 2H 2 O - 4eà O2+4H+
2H+ + 2eà H 2 2
2H2O-4eà O 2 + 4H+1
4H+ + 2H2Oà 2H 2 + 4H+ + O 2
2H2Oà 2H2+O2
Example 9. Electrolysis of potassium hydroxide solutionKOH.
Cathode (-)<-- K + + Oh - à anode (+)
Potassium cations will not be reduced at the cathode, since potassium is in the voltage series of metals to the left of aluminum, instead water molecules will be reduced:
2H2O + 2eà H 2 + 2OH - 4OH - -4eà 2H 2 O +O 2
cathode(-)2H2O+2eà H 2 + 2OH - 2
anode(+) 4OH - - 4eà 2H 2 O + O 2 1
4H 2 O + 4OH -à 2H 2 + 4OH - + 2H 2 O + O 2
2 H 2 Oà 2 H 2 + O 2
Example 10 Electrolysis of potassium nitrate solutionKNO 3 .
Cathode (-) <-- K + + NO 3 - à anode (+)
2H2O + 2eà H 2 + 2OH - 2H 2 O - 4eà O2+4H+
cathode(-)2H2O+2eà H 2 + 2OH-2
anode(+) 2H 2 O - 4eà O 2 + 4H+1
4H2O + 2H2Oà 2H2+4OH-+4H++ O2
2H2Oà 2H2+O2
When an electric current is passed through solutions of oxygen-containing acids, alkalis and salts of oxygen-containing acids with metals that are in the voltage series of metals, to the left of aluminum, water electrolysis practically occurs. In this case, hydrogen is released at the cathode, and oxygen at the anode.
Conclusions. When determining the products of electrolysis of aqueous solutions of electrolytes, in the simplest cases, one can be guided by the following considerations:
1. Metal ions with a small algebraic value of the standard potential - fromLi + beforeAl 3+ inclusive - have a very weak tendency to reattach electrons, yielding in this respect to ionsH + (cm. Cation activity series). In the electrolysis of aqueous solutions of compounds containing these cations, the function of an oxidizing agent on the cathode is performed by ionsH + , while restoring according to the scheme:
2 H 2 O+ 2 eà H 2 + 2OH -
2. Metal cations with positive values of standard potentials (Cu 2+ , Ag + , hg 2+ etc.) have a greater tendency to attach electrons than ions. During the electrolysis of aqueous solutions of their salts, these cations emit the function of an oxidizing agent on the cathode, while being reduced to a metal according to the scheme, for example:
Cu 2+ +2 eà Cu 0
3. During the electrolysis of aqueous solutions of metal saltsZn, Fe, CD, Niand others, occupying a middle position between the listed groups in the voltage series, the reduction process at the cathode occurs according to both schemes. The mass of the released metal does not correspond in these cases to the amount of electric current flowing, part of which is spent on the formation of hydrogen.
4. In aqueous solutions of electrolytes, monatomic anions (Cl - , Br - , J - ), oxygen-containing anions (NO 3 - , SO 4 2- , PO 4 3- and others), as well as hydroxyl ions of water. Of these, halide ions have the stronger reducing properties, with the exception ofF. ionsOhoccupy an intermediate position between them and polyatomic anions. Therefore, during the electrolysis of aqueous solutionsHCl, HBr, HJor their salts on the anode, halide ions are oxidized according to the scheme:
2 X - -2 eà X 2 0
During the electrolysis of aqueous solutions of sulfates, nitrates, phosphates, etc. the function of the reducing agent is performed by ions, while being oxidized according to the scheme:
4 HOH – 4 eà 2 H 2 O + O 2 + 4 H +
.
Tasks.
W a dacha 1. During the electrolysis of a solution of copper sulfate, 48 g of copper was released at the cathode. Find the volume of gas released at the anode and the mass of sulfuric acid formed in the solution.
Copper sulfate in solution dissociates neither ionsC 2+ andS0 4 2 ".
CuS0 4 \u003d Cu 2+ + S0 4 2 "
Let us write down the equations of the processes occurring at the cathode and anode. Cu cations are reduced at the cathode, electrolysis of water occurs at the anode:
Cu 2+ + 2e- \u003d Cu12
2H 2 0-4e- = 4H + + 0 2 |1
General electrolysis equation:
2Cu2+ + 2H2O = 2Cu + 4H+ + O2 (short ionic equation)
Add to both sides of the equation 2 sulfate ions each, which are formed during the dissociation of copper sulfate, we get the complete ionic equation:
2Cu2+ + 2S042" + 2H20 = 2Cu + 4H+ + 2SO4 2" + O2
2CuSO4 + 2H2O = 2Cu + 2H2SO4 + O2
The gas released at the anode is oxygen. Sulfuric acid is formed in the solution.
The molar mass of copper is 64 g / mol, we calculate the amount of copper substance:
According to the reaction equation, when 2 mol of copper is released from the anode, 1 mol of oxygen is released. 0.75 mol of copper was released at the cathode, let x mol of oxygen be released at the anode. Let's make a proportion:
2/1=0.75/x, x=0.75*1/2=0.375mol
0.375 mol of oxygen was released at the anode,
v(O2) = 0.375 mol.
Calculate the volume of released oxygen:
V(O2) \u003d v (O2) "VM \u003d 0.375 mol" 22.4 l / mol \u003d 8.4 l
According to the reaction equation, when 2 mol of copper is released at the cathode, 2 mol of sulfuric acid is formed in the solution, which means that if 0.75 mol of copper is released at the cathode, then 0.75 mol of sulfuric acid is formed in the solution, v (H2SO4) = 0.75 mol . Calculate the molar mass of sulfuric acid:
M(H2SO4) = 2-1+32+16-4 = 98 g/mol.
Calculate the mass of sulfuric acid:
m (H2S04) \u003d v (H2S04> M (H2S04) \u003d \u003d 0.75 mol \u003d 98 g / mol \u003d 73.5 g.
Answer: 8.4 liters of oxygen were released at the anode; 73.5 g of sulfuric acid was formed in the solution
Task 2. Find the volume of gases released at the cathode and anode during the electrolysis of an aqueous solution containing 111.75 g of potassium chloride. What substance is formed in solution? Find its mass.
Potassium chloride in solution dissociates into K+ and Cl ions:
2KS1 \u003d K + + Cl
Potassium ions are not reduced at the cathode; instead, water molecules are reduced. Chloride ions are oxidized at the anode and chlorine is released:
2H2O + 2e "= H2 + 20H-|1
2SG-2e "= C12|1
General electrolysis equation:
2CHl + 2H2O \u003d H2 + 2OH "+ C12 (short ionic equation) The solution also contains K + ions formed during the dissociation of potassium chloride and not participating in the reaction:
2K+ + 2Cl + 2H20 = H2 + 2K+ + 2OH" + C12
Let's rewrite the equation in molecular form:
2KS1 + 2H2O = H2 + C12 + 2KOH
Hydrogen is released at the cathode, chlorine is released at the anode, and potassium hydroxide is formed in solution.
The solution contained 111.75 g of potassium chloride.
Calculate the molar mass of potassium chloride:
M(KC1) = 39+35.5 = 74.5 g/mol
Calculate the amount of potassium chloride substance:

According to the reaction equation, electrolysis of 2 mol of potassium chloride releases 1 mol of chlorine. Let the electrolysis of 1.5 mol of potassium chloride release x mol of chlorine. Let's make a proportion:
2/1=1.5/x, x=1.5 /2=0.75 mol
0.75 mol of chlorine will be released, v (C! 2) \u003d 0.75 mol. According to the reaction equation, when 1 mol of chlorine is released at the anode, 1 mol of hydrogen is released at the cathode. Therefore, if 0.75 mol of chlorine is released at the anode, then 0.75 mol of hydrogen is released at the cathode, v(H2) = 0.75 mol.
Let us calculate the volume of chlorine released at the anode:
V (C12) \u003d v (Cl2) -VM \u003d 0.75 mol \u003d 22.4 l / mol \u003d 16.8 l.
The volume of hydrogen is equal to the volume of chlorine:
Y (H2) \u003d Y (C12) \u003d 16.8 l.
According to the reaction equation, during the electrolysis of 2 mol of potassium chloride, 2 mol of potassium hydroxide is formed, which means that during the electrolysis of 0.75 mol of potassium chloride, 0.75 mol of potassium hydroxide is formed. Calculate the molar mass of potassium hydroxide:
M (KOH) \u003d 39 + 16 + 1 - 56 g / mol.
Calculate the mass of potassium hydroxide:
m(KOH) \u003d v (KOH> M (KOH) \u003d 0.75 mol-56 g / mol \u003d 42 g.
Answer: 16.8 liters of hydrogen were released at the cathode, 16.8 liters of chlorine were released at the anode, and 42 g of potassium hydroxide formed in the solution.
Task 3. During the electrolysis of a solution of 19 g of divalent metal chloride at the anode, 8.96 liters of chlorine were released. Determine which metal chloride was subjected to electrolysis. Calculate the volume of hydrogen released at the cathode.
We denote the unknown metal M, the formula of its chloride is MC12. At the anode, chloride ions are oxidized and chlorine is released. The condition says that hydrogen is released at the cathode, therefore, water molecules are reduced:
2H20 + 2e- = H2 + 2OH|1
2Cl -2e "= C12! 1
General electrolysis equation:
2Cl + 2H2O \u003d H2 + 2OH "+ C12 (short ionic equation)
The solution also contains M2+ ions, which do not change during the reaction. We write the full ionic reaction equation:
2SG + M2+ + 2H2O = H2 + M2+ + 2OH- + C12
Let's rewrite the reaction equation in molecular form:
MS12 + 2H2O - H2 + M(OH)2 + C12
Find the amount of chlorine released at the anode:
![]()
According to the reaction equation, during the electrolysis of 1 mol of chloride of an unknown metal, 1 mol of chlorine is released. If 0.4 mol of chlorine was released, then 0.4 mol of metal chloride was subjected to electrolysis. Calculate the molar mass of metal chloride:
The molar mass of chloride of an unknown metal is 95 g/mol. There are 35.5"2 = 71 g/mol per two chlorine atoms. Therefore, the molar mass of the metal is 95-71 = 24 g/mol. Magnesium corresponds to this molar mass.
According to the reaction equation, for 1 mole of chlorine released at the anode, there is 1 mole of hydrogen released at the cathode. In our case, 0.4 mol of chlorine was released at the anode, which means that 0.4 mol of hydrogen was released at the cathode. Calculate the volume of hydrogen:
V (H2) \u003d v (H2> VM \u003d 0.4 mol \u003d 22.4 l / mol \u003d 8.96 l.
Answer: subjected to electrolysis solution of magnesium chloride; 8.96 liters of hydrogen were released at the cathode.
*Problem 4. During the electrolysis of 200 g of a solution of potassium sulfate with a concentration of 15%, 14.56 liters of oxygen were released at the anode. Calculate the concentration of the solution at the end of the electrolysis.
In a solution of potassium sulfate, water molecules react both at the cathode and at the anode:
2H20 + 2e "= H2 + 20H-|2
2H2O - 4e "= 4H+ + O2! 1
Let's put both equations together:
6H2O \u003d 2H2 + 4OH "+ 4H + + O2, or
6H2O \u003d 2H2 + 4H2O + O2, or
2H2O = 2H2 + 02
In fact, during the electrolysis of a solution of potassium sulfate, the electrolysis of water occurs.
The concentration of a solute in a solution is determined by the formula:
C=m(solute) 100% / m(solution)
To find the concentration of the potassium sulfate solution at the end of the electrolysis, it is necessary to know the mass of potassium sulfate and the mass of the solution. The mass of potassium sulfate does not change during the reaction. Calculate the mass of potassium sulfate in the initial solution. Let us denote the concentration of the initial solution as C
m(K2S04) = C2 (K2S04) m(solution) = 0.15 200 g = 30 g.
The mass of the solution changes during electrolysis, as part of the water is converted into hydrogen and oxygen. Calculate the amount of released oxygen:
(O 2) \u003d V (O2) / Vm \u003d 14.56 l / 22.4 l / mol \u003d 0.65 mol
According to the reaction equation, 1 mole of oxygen is formed from 2 moles of water. Let 0.65 mol of oxygen be released during the decomposition of x mol of water. Let's make a proportion:
![]()
1.3 mol of water decomposed, v(H2O) = 1.3 mol.
Calculate the molar mass of water:
M(H2O) \u003d 1-2 + 16 \u003d 18 g / mol.
Calculate the mass of decomposed water:
m(H2O) \u003d v (H2O> M (H2O) \u003d 1.3 mol * 18 g / mol \u003d 23.4 g.
The mass of the potassium sulfate solution decreased by 23.4 g and became equal to 200-23.4 = 176.6 g. Let us now calculate the concentration of the potassium sulfate solution at the end of the electrolysis:
С2 (K2 SO4)=m(K2 SO4) 100% / m(solution)=30g 100% / 176.6g=17%
Answer: the concentration of the solution at the end of the electrolysis is 17%.
* 3 problem 5. 188.3 g of a mixture of sodium and potassium chlorides were dissolved in water and an electric current was passed through the resulting solution. During electrolysis, 33.6 liters of hydrogen were released at the cathode. Calculate the composition of the mixture in percent by weight.
After dissolving a mixture of potassium and sodium chlorides in water, the solution contains K+, Na+ and Cl- ions. Neither potassium ions nor sodium ions are reduced at the cathode, water molecules are reduced. Chloride ions are oxidized at the anode and chlorine is released:
Let's rewrite the equations in molecular form:
2KS1 + 2H20 = H2 + C12 + 2KOH
2NaCl + 2H2O = H2 + C12 + 2NaOH
Let us denote the amount of potassium chloride substance contained in the mixture, x mol, and the amount of sodium chloride substance, y mol. According to the reaction equation, during the electrolysis of 2 mol of sodium or potassium chloride, 1 mol of hydrogen is released. Therefore, during electrolysis x mol of potassium chloride, x / 2 or 0.5x mol of hydrogen is formed, and during electrolysis, y mol of sodium chloride is 0.5y mol of hydrogen. Let's find the quantity hydrogen substances, released during the electrolysis of the mixture:
![]()
Let's make the equation: 0.5x + 0.5y \u003d 1.5
Calculate the molar masses of potassium and sodium chlorides:
M(KC1) = 39+35.5 = 74.5 g/mol
M (NaCl) \u003d 23 + 35.5 \u003d 58.5 g / mol
Mass x mole of potassium chloride is:
m (KCl) \u003d v (KCl) -M (KCl) \u003d x mol-74.5 g / mol \u003d 74.5 x g.
The mass of a mole of sodium chloride is:
m (KCl) \u003d v (KCl) -M (KCl) \u003d y mol-74.5 g / mol \u003d 58.5 u g.
The mass of the mixture is 188.3 g, we make the second equation:
74.5x + 58.5y = 188.3
So, we solve a system of two equations with two unknowns:
0.5(x + y)= 1.5
74.5x + 58.5y = 188.3g
From the first equation, we express x:
x + y \u003d 1.5 / 0.5 \u003d 3,
x = 3-y
Substituting this value of x into the second equation, we get:
74.5-(3-y) + 58.5y = 188.3
223.5-74.5y + 58.5y = 188.3
-16y = -35.2
y \u003d 2.2 100% / 188.3g \u003d 31.65%
Calculate the mass fraction of sodium chloride:
w(NaCl) = 100% - w(KCl) = 68.35%
Answer: the mixture contains 31.65% potassium chloride and 68.35% sodium chloride.
When considering the electrolysis of solutions, one should not lose sight of the fact that, in addition to electrolyte ions, in any aqueous solution there are also ions that are still products of the dissociation of water - H + and OH -. In an electrolytic field, hydrogen ions move to the cathode, and hydroxyl to the anode. Thus, both electrolyte cations and hydrogen cations can be discharged at the cathode. Similarly, at the anode, both electrolyte anions and hydroxyl ions can be discharged. In addition, water molecules can also undergo electrochemical oxidation or reduction.
Which electrochemical processes will take place at the electrodes during electrolysis will primarily depend on the ratio of the electrode potentials of the corresponding electrochemical systems. This means that the oxidized forms of electrochemical systems will be reduced at the cathode. Of the several possible processes, the one with the minimum energy consumption will proceed. This means that the oxidized forms of electrochemical systems with the highest electrode potential will be reduced at the cathode, while the reduced forms of systems with the lowest electrode potential will be oxidized at the anode. The electrode material has an inhibitory effect on the course of some electrochemical processes; such cases are discussed below.
Considering the cathodic processes occurring during the electrolysis of aqueous solutions, it is necessary to take into account the value of the potential of the hydrogen ion reduction process. This potential depends on the concentration of hydrogen ions and in the case of neutral solutions (рН=7) has the value
φ \u003d -0.059 * 7 \u003d -0.41 V. From this it is clear that if the electrolyte is formed by a metal whose electrode potential is much more positive than -0.41 V, then metal will be released from the neutral solution at the cathode. Such metals are in a series of voltages near hydrogen (starting approximately from tin) and after it. On the contrary, in the case of electrolytes, the metal of which has a potential much more negative than –0.41 V, the metal will not be reduced, but hydrogen will be released. Such metals include metals of the beginning of a series of stresses, up to approximately titanium. Finally, if the potential of the metal is close to -0.41 V (metals of the middle part of the Zn, Cr, Fe, Ni series), then, depending on the solution concentration and electrolysis conditions, both metal reduction and hydrogen evolution are possible; often there is a joint release of metal and hydrogen.
The electrochemical evolution of hydrogen from acidic solutions occurs as a result of the discharge of hydrogen ions. In the case of neutral or alkaline media, it is the result of the electrochemical reduction of water:
2H 2 O + 2e - \u003d H 2 + 2OH -
Thus, the nature of the cathodic process during the electrolysis of aqueous solutions is determined primarily by the position of the corresponding metal in the series of voltages. In some cases, the pH of the solution, the concentration of metal ions, and other electrolysis conditions are of great importance.
When considering anode processes, it should be borne in mind that the anode material can oxidize during electrolysis. In this regard, electrolysis with an inert anode and electrolysis with an active anode are distinguished. An anode is called inert, the material of which does not undergo oxidation during electrolysis. The active anode is the anode, the material of which can be oxidized during electrolysis. As materials for inert anodes, graphite, coal, and platinum are more often used.
On an inert anode during the electrolysis of aqueous solutions of alkalis, oxygen-containing acids and their salts, as well as hydrofluoric acid and fluorides, electrochemical oxidation of water occurs with the release of oxygen. Depending on the pH of the solution, this process proceeds differently and can be written in different equations. AT alkaline environment the equation looks like
4OH - \u003d O 2 + 2H 2 O + 4e -
and in acidic or neutral:
2H 2 O \u003d O 2 + 4H + + 4e -
In the cases under consideration, the electrochemical oxidation of water is the most energetically favorable process. Oxygen-containing anions are either not able to be oxidized, or their oxidation occurs at very high potentials. For example, the standard oxidation potential of the SO 4 2- ion
2SO 4 2- \u003d S 2 O 8 2- + 2e -
is 2.010 V, which is much higher than the standard water oxidation potential (1.228 V). The standard oxidation potential of the F ion is even greater (2.87 V).
During the electrolysis of aqueous solutions of anoxic acids and their salts (except for HF and fluorides), anions are discharged at the anode. In particular, during the electrolysis of solutions of HI, HBr, HCl and their salts, the corresponding halogen is released at the anode. Note that the release of chlorine during the electrolysis of HCl and its salts contradicts the mutual position of the systems
2Cl - \u003d 2Cl + 2e - (φ \u003d 1.359 V)
2H 2 O \u003d O 2 + 4H + + 4e - (φ \u003d 1.228 V)
in a series of standard electrode potentials. This anomaly is associated with a significant overvoltage of the second of these two electrode processes - the anode material has an inhibitory effect on the process of oxygen evolution.
In the case of an active anode, the number of competing oxidative processes increases to three: electrochemical oxidation of water with oxygen evolution, anion discharge (i.e., its oxidation), and electrochemical oxidation of the anode metal (the so-called anodic dissolution of the metal). Of these possible processes, the one that is energetically most favorable will proceed. If the anode metal is located in a series of standard potentials earlier than both other electrochemical systems, then anodic dissolution of the metal will be observed. Otherwise, there will be an evolution of oxygen or an anion discharge.
Let us consider several typical cases of electrolysis of aqueous solutions.
Electrolysis of a CuCl 2 solution with an inert anode. Copper in the series of voltages is located after hydrogen; therefore, at the cathode there will be a discharge of Cu 2+ ions and the release of metallic copper. At the anode, chloride ions will be discharged.
Scheme of electrolysis of a solution of chloride stranded (II):
Cathode ← Cu 2+ 2Cl - → Anode
Cu 2+ + 2e - \u003d Cu 2Cl - \u003d 2Cl + 2e -
Electrolysis of K 2 SO 4 solution with an inert anode. Since potassium in the series of voltages is much earlier than hydrogen, hydrogen will be released at the cathode and OH - will accumulate. At the anode, oxygen will be released and H + ions will accumulate. At the same time, K + ions will come into the cathode space, and SO 4 2- ions into the anode space. Thus, the solution in all its parts will remain electrically neutral. However, alkali will accumulate in the cathode space, and acid will accumulate in the anodic space.
Scheme of electrolysis of potassium sulfate solution:
Cathode ← 4K + 2SO 4 2- → Anode
4H 2 O + 4e - \u003d 4OH - + 4H 2H 2 O \u003d 4H + + 2O + 4e -
KOH 4H \u003d 2H 2 2O \u003d O 2 H 2 SO 4
Electrolysis of NiSO 4 solution with nickel anode. The standard potential of nickel (-0.250 V) is somewhat greater than -0.41 V; therefore, during the electrolysis of a neutral solution of NiSO 4 on the cathode, the Ni 2+ ions are mainly discharged and the metal is released. At the anode, the opposite process occurs - the oxidation of the metal, since the nickel potential is much less than the oxidation potential of water, and even more so, the oxidation potential of the SO 4 2- ion. Thus, in this case, electrolysis is reduced to the dissolution of the anode metal and its separation at the cathode.
Nickel sulfate solution electrolysis scheme:
Cathode ← Ni 2+ SO 4 2- → Anode
Ni 2+ + 2e - \u003d Ni Ni \u003d Ni 2+ + 2e -
This process is used for the electrochemical purification of nickel.
Faraday's laws
1. Faraday's law.
The mass of the substance released on the electrode when an electric current passes through the electrolyte solution is directly proportional to the amount of electricity.
Where ∆m is the amount of reacted substance; Q is the amount of electricity; k e - coefficient of proportionality, showing how much the substance reacted during the passage of a unit of electricity. The value, k is called the electrochemical equivalent.
k=M/(N A z│e│)
where z is the ion valency; M is the molar mass of the substance released on the electrode; N A is the Avogadro constant. │e│= 1.6 10 -19 Cl.
2. Faraday's law.
According to Faraday's second law, with a certain amount of electricity passed, the ratio of the masses of the reacted substances is equal to the ratio of their chemical equivalents:
∆m 1 /A 1 =∆m 2 /A 2 =∆m 3 /A 3 = const
The chemical equivalent of an element is equal to the ratio of the part of the mass of the element that adds or replaces in chemical compounds one atomic mass of hydrogen or half an atomic mass of oxygen, to 1/12 of the mass of a C 12 atom. The concept of “chemical equivalent” is also applicable to compounds. Thus, the chemical equivalent of an acid is numerically equal to its molar mass divided by the basicity (number of hydrogen ions), the chemical equivalent of a base is its molar mass divided by acidity (for an inorganic base, by the number of hydroxyl groups), the chemical equivalent of a salt is its molar mass, divided by the sum of the charges of the cations or anions.
What is electrolysis? For a simpler understanding of the answer to this question, let's imagine any source of direct current. For every DC source, you can always find a positive and a negative pole:
Let us connect to it two chemically resistant electrically conductive plates, which we will call electrodes. The plate connected to the positive pole is called the anode, and to the negative pole is called the cathode:
Sodium chloride is an electrolyte; when it melts, it dissociates into sodium cations and chloride ions:
NaCl \u003d Na + + Cl -
It is obvious that the negatively charged chlorine anions will go to the positively charged electrode - the anode, and the positively charged Na + cations will go to the negatively charged electrode - the cathode. As a result of this, both Na + cations and Cl - anions will be discharged, that is, they will become neutral atoms. The discharge occurs through the acquisition of electrons in the case of Na + ions and the loss of electrons in the case of Cl − ions. That is, the process proceeds at the cathode:
Na + + 1e − = Na 0 ,
And on the anode:
Cl − − 1e − = Cl
Since each chlorine atom has an unpaired electron, their single existence is unfavorable and the chlorine atoms combine into a molecule of two chlorine atoms:
Сl∙ + ∙Cl \u003d Cl 2
Thus, in total, the process occurring at the anode is more correctly written as follows:
2Cl - - 2e - = Cl 2
That is, we have:
Cathode: Na + + 1e − = Na 0
Anode: 2Cl - - 2e - = Cl 2
Let's sum up the electronic balance:
Na + + 1e − = Na 0 |∙2
2Cl − − 2e − = Cl 2 |∙1<
Add the left and right sides of both equations half reactions, we get:
2Na + + 2e − + 2Cl − − 2e − = 2Na 0 + Cl 2
We reduce two electrons in the same way as it is done in algebra, we get the ionic equation of electrolysis:
2NaCl (l.) => 2Na + Cl 2
From a theoretical point of view, the case considered above is the simplest, since in the sodium chloride melt, among the positively charged ions, there were only sodium ions, and among the negative ones, only chlorine anions.
In other words, neither Na + cations nor Cl − anions had "competitors" for the cathode and anode.
And what will happen, for example, if instead of a melt of sodium chloride, a current is passed through its aqueous solution? Dissociation of sodium chloride is also observed in this case, but the formation of metallic sodium in an aqueous solution becomes impossible. After all, we know that sodium, a representative of alkali metals, is an extremely active metal that reacts very violently with water. If sodium cannot be reduced under such conditions, then what will be reduced at the cathode?
Let's remember the structure of the water molecule. It is a dipole, that is, it has a negative and a positive pole:
It is due to this property that it is able to “stick around” both the cathode surface and the anode surface:
The following processes may take place:
2H 2 O + 2e - \u003d 2OH - + H 2
2H 2 O - 4e - \u003d O 2 + 4H +
Thus, it turns out that if we consider a solution of any electrolyte, we will see that the cations and anions formed during the dissociation of the electrolyte compete with water molecules for reduction at the cathode and oxidation at the anode.
So what processes will take place at the cathode and at the anode? Discharge of ions formed during the dissociation of the electrolyte or oxidation / reduction of water molecules? Or, perhaps, all of these processes will occur simultaneously?
Depending on the type of electrolyte, a variety of situations are possible during the electrolysis of its aqueous solution. For example, cations of alkali, alkaline earth metals, aluminum and magnesium are simply not able to be reduced in the aquatic environment, since their reduction should have produced alkali, alkaline earth metals, aluminum or magnesium, respectively. metals that react with water.
In this case, only the reduction of water molecules at the cathode is possible.
It is possible to remember what process will take place on the cathode during the electrolysis of a solution of any electrolyte, following the following principles:
1) If the electrolyte consists of a metal cation, which in a free state in normal conditions reacts with water, the process is going on at the cathode:
2H 2 O + 2e - \u003d 2OH - + H 2
This applies to metals that are at the beginning of the Al activity series, inclusive.
2) If the electrolyte consists of a metal cation, which in its free form does not react with water, but reacts with non-oxidizing acids, two processes take place at once, both the reduction of metal cations and water molecules:
Me n+ + ne = Me 0
These metals include those between Al and H in the activity series.
3) If the electrolyte consists of hydrogen cations (acid) or metal cations that do not react with non-oxidizing acids, only electrolyte cations are restored:
2H + + 2e - \u003d H 2 - in the case of acid
Me n + + ne = Me 0 - in the case of salt
At the anode, meanwhile, the situation is as follows:
1) If the electrolyte contains anions of oxygen-free acid residues (except F -), then the process of their oxidation takes place at the anode, water molecules are not oxidized. For example:
2Cl - - 2e \u003d Cl 2
S 2- − 2e = S o
Fluoride ions are not oxidized at the anode because fluorine is not able to form in an aqueous solution (reacts with water)
2) If the electrolyte contains hydroxide ions (alkalis), they are oxidized instead of water molecules:
4OH - - 4e - \u003d 2H 2 O + O 2
3) If the electrolyte contains an oxygen-containing acid residue (except for organic acid residues) or a fluoride ion (F -) on the anode, the process of oxidizing water molecules takes place:
2H 2 O - 4e - \u003d O 2 + 4H +
4) In the case of an acidic residue of a carboxylic acid on the anode, the following process takes place:
2RCOO - - 2e - \u003d R-R + 2CO 2
Let's practice writing electrolysis equations for various situations:
Example #1
Write the equations for the processes occurring at the cathode and anode during the electrolysis of a zinc chloride melt, as well as the general electrolysis equation.
Solution
When zinc chloride is melted, it dissociates:
ZnCl 2 \u003d Zn 2+ + 2Cl -
Further, attention should be paid to the fact that it is the zinc chloride melt that undergoes electrolysis, and not the aqueous solution. In other words, without options, only the reduction of zinc cations can occur at the cathode, and the oxidation of chloride ions at the anode. no water molecules
Cathode: Zn 2+ + 2e − = Zn 0 |∙1
Anode: 2Cl − − 2e − = Cl 2 |∙1
ZnCl 2 \u003d Zn + Cl 2
Example #2
Write the equations for the processes occurring at the cathode and anode during the electrolysis of an aqueous solution of zinc chloride, as well as the general electrolysis equation.
Since in this case, an aqueous solution is subjected to electrolysis, then, theoretically, water molecules can take part in electrolysis. Since zinc is located in the activity series between Al and H, this means that both the reduction of zinc cations and water molecules will occur at the cathode.
2H 2 O + 2e - \u003d 2OH - + H 2
Zn 2+ + 2e − = Zn 0
The chloride ion is the acidic residue of the oxygen-free acid HCl, therefore, in the competition for oxidation at the anode, chloride ions “win” over water molecules:
2Cl - - 2e - = Cl 2
In this particular case, it is impossible to write the overall electrolysis equation, since the ratio between hydrogen and zinc released at the cathode is unknown.
Example #3
Write the equations for the processes occurring at the cathode and anode during the electrolysis of an aqueous solution of copper nitrate, as well as the general electrolysis equation.
Copper nitrate in solution is in a dissociated state:
Cu(NO 3) 2 \u003d Cu 2+ + 2NO 3 -
Copper is in the activity series to the right of hydrogen, that is, copper cations will be reduced at the cathode:
Cu 2+ + 2e − = Cu 0
Nitrate ion NO 3 - is an oxygen-containing acid residue, which means that in oxidation at the anode, nitrate ions “lose” in competition with water molecules:
2H 2 O - 4e - \u003d O 2 + 4H +
In this way:
Cathode: Cu 2+ + 2e − = Cu 0 |∙2
2Cu 2+ + 2H 2 O = 2Cu 0 + O 2 + 4H +
The equation obtained as a result of addition is the ionic equation of electrolysis. To get the complete molecular electrolysis equation, you need to add 4 nitrate ions to the left and right sides of the resulting ionic equation as counterions. Then we will get:
2Cu(NO 3) 2 + 2H 2 O = 2Cu 0 + O 2 + 4HNO 3
Example #4
Write the equations for the processes occurring at the cathode and anode during the electrolysis of an aqueous solution of potassium acetate, as well as the general electrolysis equation.
Solution:
Potassium acetate in an aqueous solution dissociates into potassium cations and acetate ions:
CH 3 COOK \u003d CH 3 COO − + K +
Potassium is an alkali metal, i.e. is in the electrochemical series of voltages at the very beginning. This means that its cations are not capable of being discharged at the cathode. Instead, water molecules will be restored:
2H 2 O + 2e - \u003d 2OH - + H 2
As mentioned above, the acid residues of carboxylic acids “win” in the competition for oxidation from water molecules at the anode:
2CH 3 COO - - 2e - \u003d CH 3 -CH 3 + 2CO 2
Thus, summing up the electronic balance and adding the two equations of half-reactions at the cathode and anode, we obtain:
Cathode: 2H 2 O + 2e − = 2OH − + H 2 |∙1
Anode: 2CH 3 COO - - 2e - \u003d CH 3 -CH 3 + 2CO 2 | ∙ 1
2H 2 O + 2CH 3 COO - \u003d 2OH - + H 2 + CH 3 -CH 3 + 2CO 2
We have obtained the complete electrolysis equation in ionic form. By adding two potassium ions to the left and right sides of the equation and adding them with counterions, we get the complete electrolysis equation in molecular form:
2H 2 O + 2CH 3 COOK \u003d 2KOH + H 2 + CH 3 -CH 3 + 2CO 2
Example #5
Write the equations for the processes occurring at the cathode and anode during the electrolysis of an aqueous solution of sulfuric acid, as well as the general electrolysis equation.
Sulfuric acid dissociates into hydrogen cations and sulfate ions:
H 2 SO 4 \u003d 2H + + SO 4 2-
Hydrogen cations H + will be reduced at the cathode, and water molecules will be oxidized at the anode, since sulfate ions are oxygen-containing acid residues:
Cathode: 2Н + + 2e − = H 2 |∙2
Anode: 2H 2 O - 4e - = O 2 + 4H + |∙1
4H + + 2H 2 O \u003d 2H 2 + O 2 + 4H +
Reducing the hydrogen ions in the left and right and left sides of the equation, we obtain the equation for the electrolysis of an aqueous solution of sulfuric acid:
2H 2 O \u003d 2H 2 + O 2
As can be seen, the electrolysis of an aqueous solution of sulfuric acid is reduced to the electrolysis of water.
Example #6
Write the equations for the processes occurring at the cathode and anode during the electrolysis of an aqueous solution of sodium hydroxide, as well as the general electrolysis equation.
Dissociation of sodium hydroxide:
NaOH = Na + + OH -
Only water molecules will be reduced at the cathode, since sodium is a highly active metal, and only hydroxide ions at the anode:
Cathode: 2H 2 O + 2e − = 2OH − + H 2 |∙2
Anode: 4OH − − 4e − = O 2 + 2H 2 O |∙1
4H 2 O + 4OH - \u003d 4OH - + 2H 2 + O 2 + 2H 2 O
Let us reduce two water molecules on the left and on the right and 4 hydroxide ions and come to the conclusion that, as in the case of sulfuric acid, the electrolysis of an aqueous solution of sodium hydroxide is reduced to the electrolysis of water.